Commentry - (2017) Volume 3, Issue 1
Integration of Molecular Methods into Microbiological Diagnostics
*Corresponding Author: Ousman Bajinka, Department of Biology, Ege University, Inciralti Ogrinci Yurt, İnciraltı Mh. 35340 Balçova/İzmir, Turkey, Tel: + 5061792543, Fax: + 5061792543 Email:
Abstract
Conventional microbiological methods take a long time to complete and sometimes accuracy can be compromised due to varying levels of expertise in the laboratory. Thus, molecular methods are highly needed to accomplish this mission. Apart from the specificity and sensitivity, molecular methods confer accuracy, precision, reproductive among others.
This review points out the respective needs for molecular techniques in diagnostic laboratories. Various convincing points were elaborated including; the short turnaround time, minimization of nosocomial infections (transmission within the community and health care system), the economic cost involved in-patient treatment, the sensitivity, reliability and the accurate diagnosis of infectious diseases.
The benefits that epidemiological studies draw from molecular techniques need to be implemented in developing nations, the big hopes, some limitations and recommendations of the use molecular methods in microbiological diagnostics are discussed.
Keywords: Infectious diseases; Molecular methods; Nosocomial infection; Microbial diagnosis
Introduction
The need for molecular methods in the microbiological diagnostics of infectious diseases has come of age. The reasons are based on; the timely advantages, detection of slow growing pathogen or nonculturable organisms that are more or less difficult to detect, identify and even to be test for antimicrobial susceptibility by conventional methods [1-5]. This century is conciding with many highly infectious pathogens, notably HIV AIDS that has been dictating our immune response to the emerging of number of infections [2,3,6-10].
Nowadays conditions such as HIV depends entirely on molecular diagnosis for susceptibility testing and management even during the acute phase [11]. In fact the hurdles that are faced when diagnosing a plethora of respiratory pathogens can now be accurately achieved even in a simpler and quicker manner than that offered by conventional approaches [1,10]. In the recent past, the replacement of traditional methods by molecular methods was thought to be far from achievable but with an increase in variety of commercially available kits, these hopes have now been reinstated [12]. Table 1 that shows molecular testing winning the upper hand against the culture method.
| Culture | Molecualr | |
|---|---|---|
| Sensitivity | Fair | 3 to 5 fold greater than culture |
| Specificity | Very High | Very High |
| Vulnerability to transport promblems | Sensitive to extreme temperatures and drying | Viable specimen not required |
| Typing | 2ndstep often required | Incorporated in initial Procedure |
| Turn around Time | 2-3 days | 4-8 h |
Table 1: Comparing culture and molecular testing (Felix Martinez, Jr., M.D Genital Herpes: Molecular Testing Can Increase Sensitivity For Detection of Herpes Virus, 2012).
Sensitivity of the Tests
Humans are always prone to making mistakes and these ranges from fictions to realities. However, with the invention of more sophisticated machines a considerable reduction in human error have been assured. From conventional diagnostic to molecular methods, conventional PCR to real time PCR, from singleplex to multiplex PCR and from this stage to the promising high-throughput sequencing (HTS). With all of these, the purpose of achieving one condition will never be defeated; the sensitivity that molecular techniques has to offer. With the advance of techniques like; microfluidics, mutliplex with its Taqman probe system and information processing, sensitivity will always stands despensable (Table 2) [13].
| Technique | Sensitivity | Specificity | Advantages | Disavantages |
|---|---|---|---|---|
| Culture Methods | 50% | 100% | Gold standard: Allows susceptabilities | Delay in Diagnosis: Low sensitivity |
| PCR Methods | 90% | 100% | Highest accuracy | Low commercial availability |
| ß-D-glucan | 70-100% | 87-96% | Panfungal marker: high sensitivity and specificity | many false-positive results;methodological concerns |
| Galactomannan | 80-90% | 80-90% | Increased accuracy for detection of Aspergillus in hematologic illnesses | Only for Aspergillus;many false-positive results; not useful in hematologic illnesses |
| Mannan or Antimannan | 60-89% | 80-84% | Good specificity and sensitivity with combined use | Results vary: limited experience |
| combination | 87% | 84% |
Table 2: Accuracy of diagnostic testing for invasive fungal infectionsin the ICU.
Apart from the early detection of Mycobacterium tuberculosis, Chlamydia trachomatis, meningo-encephalitis syndrome and respiratory infections, the use of molecular methods also confer accurate diagnosis [1,3]. In addition to the different PCR’s techniques, sanger sequencing, pryosequencing, mass spectrometry, microarray analysis and reverse hybridization, the molecular diagnosis has established an advanced blood culture techniques for identification and antimicrobial susceptibility tests (Antimicrobial susceptibility tests are commonly used to determine which specific antibiotic is sensitive to a pathogen). Nowadays a specific diagnosis and a concret decision can be for specific treatment options in the mangament of infectious diseases (Table 3) [2,13].
| Inoculum Shigella | Multiplex PCR Positive results | Culture Positive results | |
|---|---|---|---|
| (CFU/Sample) | Enriched PCR Samples | Direct PCR samples | |
| 107 | 3/3 | 0/3 | 10/10 |
| 106 | 3/3 | 0/3 | 5/10 |
| 105 | 10/10 | 0/3 | 1/10 |
| 104 | 10/10 | 0/3 | 0/3 |
| 103 | 0/10 | 0/3 | 0/3 |
| 102 | 0/3 | 0/3 | 0/3 |
| 10 | 0/3 | 0/3 | 0/3 |
| Non Inoculated | 0/10 | 0/3 | 0/10 |
Table 3: Detection level of the PCR assay (Direct and Enriched) ascompared to the conventional culture, using serial dilutions of Shigella flexneri spiked to lettuce samples.
Reliability of the Results
Molecular techniques has added an exceptional discriminating power that confers a more reliable identification among and between strains of pathogens with high accuracy [1.16.5]. Of course, we will have to resort to methods that are more sophisticated if traditional methods failed to produce a good result. The low sensitivity arising from microscopy for example protozoan like; Trichomonas vaginalis, Neisseria gonorrhoeae (sexual transmitted infections) and intracellular pathogens like; virus, Mycoplasma genitalium not neglecting the low sensitivity of Chlamydia sp. and Neisseria [2] has caused for an alarm that the application of more inovative and sensitive methods is highly needed. In addition, other challenging calls for concern are the seropositivity (the quality or state of being seropositive, of having blood serum that tests positive for given pathogen) in Chlamydia sp. and the very slow growth of Mycobacterium tuberculosis also catalazed the need for a more sensitive techniques [2,14]. In a paper on ‘Is There a Fungus Among Us - An Update on Diagnostic Strategies’, PCR methods shows an increased 90% sensitivity while 50% for culture methods. This automaticaly catalyst the need for molecular methods to replace the less sensitive culture testings.
Detection and Identification of Nosocomial infections
The use of molecular methods will also reduce the risk of transmission within the community and health care system nosocomial infections [1,4]. Apart from its health complications, nosocomial infections also cause an increase in the cost of intensive care and prolong hospitalization by weeks or more [4]. Although with conventional methods, the study of epidemiology on nosocomial infections is next to impossible, with the invention of the molecular methods of strain typing; a positive difference have been seen [4,14]. The introduction of molecular biology methods that can detect DNA and RNA (nucleic acid probing and amplification) will no doubt help in strain typing for the types of nosocomial occuring at a particular time [1,6].
In Epidemiological Studies
Epidemiological studies are important to understand the transmission mechanisms and the role of microbial variants in the spread of diseases in society [15]. In the past decades, epidemiological studies were based on only phenotypical identification and for this, strain typing is not always possible [1]. The need for reference-based on data in the diagnosis has been highlighted through the use of nucleic acid amplification techniques [4]. And this has given a greater room for improvement in scientific discoveries [1]. Understanding the antimicrobial susceptility profiles for an organism is an important first step for studying epidemiology [7,15].
Improved Laboratory Turn-around Time (TAT)
The detection and identification of pathogens in a clinical laboratory is traditionally done by the microscopy, culture and sensitivity (MC&S).The sample can be view under microscope as the first stem that can quickly enable the clinician to decide on the commencement of the treatment based on the gram type. This is normally followed by culturing of the pathogen that passes through sub cultural and hence colony count is possible. Moreover, a pure culture is then subjected to a disc contining agar with antibiotics that can show the resistant and sensitive pathogens. These proceudures are loborious and time consuming [1.10.14] However when pathogen(s) can be detected in few hours after a patient visit the hospital, there is always an improvement and patient care is ensured [1,6,16,17]. Culture based methods can take days and sometimes weeks before the discrimination of sample [1,12]. And, to ensure pure isolation, sub culture causes further delays [10]. In order to create a boundary for the morbidity and mortality rate that Invasive Fungal Infections (IFIs) are causing, early and acurate diagnosis is the key [8,9]. The clinical decisions made due to results obtained from the use of conventional methods, histological and radiological techniques in mycology are less rapid, sensitive and accurate when compared to molecular techniques (Table 4) [8]. Molecular methods can quickly establish antifungal therapy and avoid the toxic production by the antifungal agents in a more reliable manner [8,9].
| Detection Method | Food borne Pathogens | Detection limit | Food matrix | Assay time |
|---|---|---|---|---|
| Multiplex PCR | Salmonella spp., Salmonella enteritidis | 103CFU/mL | Artificially and naturally contaminated chicken carcasses, minas cheese and fresh pork sausages | 24 h |
| STEC O26, O103, O111, O145, sorbitol fermenting O157, non-sorbitol fermenting O157 | 5 × 104CFU/mL in minced beef and sprouted seeds. 5 × 103CFU/mL in raw milk cheese | Artcificaillycontaminated minced beef, sprouted seeds (soy, alfafa and leek) and raw milk cheese | 24 h | |
| Escherichia coli O157: H7, Listeria monocytogenes, Staphylococcus aureus, Yersinia enterocolitica, Salmonella | 103CFU/ mL | Artificially contaminated pork | Not stated | |
| Real-time PCR | Salmonella enterica | 41.2 fg/PCR for Salmonella typhimurium genomic DNA, 18.6 fg/PCR for Salmonella enteritidis genomic DNA | Artificially contaminated chicken, liquid egg, and peanut butter | 10 h |
| Listeria monocytogenes, Escherichia coli O157, Salmonella spp | <18 CFU/10 g | Artificially contaminated ground beef, Naturally contaminated beef, pork, turkey and chicken | 24 h | |
| Listeria monocytogenes, Escherichia coli O157, Salmonella spp | 2 ×102CFU/mL | Artificially contaminated ground pork | 24 h | |
| Salmonella spp., Listeria monocytogenes | 5 CFU/25g | Artificially and naturally contaminated meat, fish, fruits, vegetables, dairy products, eggs, chocolate bar, omelet, lasagna, and various cooked dishes | <30 h | |
| Staphylococcus aureus, Salmonella, Shigella | 9.6 CFU/g for Staphylococcus aureus, 2.0 CFU/g for Salmonella and 6.8 CFU/g for Shigella | Fresh pork | <8 h | |
| NASBA | Escherichia coli | 40 cells/mL | Drinking water | 4 h |
| Salmonella enteritidis | 101 CFU/reaction | Artificially contaminated fresh meats, poultry, fish, ready-to eat salads and bakery products | 26 h | |
| Listeria monocytogenes | 400 CFU/mL | Artificially contaminated cooked ham and smoked salmon slices | 72 h | |
| Bacillus amyloliquefaciens, Bacillus cereus and Bacillus circulans | - | Artificially contaminated milk | Not stated | |
| Salmonella Enteritidis and Salmonella typhimurium | <10 CFU/mL | - | <90 min | |
| LAMP | Vibrio vulnificus | 5.4 CFU/reaction for a virulent V.vulnificusin a pure culture. 2.5 × 103CFU/g for a virulent V.vulnificusstrain in spiked raw oyster, no enrichment. 1 CFU/g for a virulent V.vulnificus strain spiked in raw oyster after 6 h enrichment | Artificially contaminated raw oysters | 8 h |
| Salmonella spp. and Shigella spp. | 5 CFU/10mL | Artificially contaminated milk | <20 h | |
| Vibrio parahaemolyticus | 10 CFU/reaction | Naturally contaminated seafood samples: fish, shrimp and mussel | 16 h | |
| STEC O26, O45, O103, O111, O121, O145, and O157 | 1-20 cells/reaction in pur culture and 105-106CFU/25g in produce | Artificially contaminated lettuce, spinach and sprouts | Not stated | |
| Oligonucleotide DNA microarray | Esherichia coliO157: H7, Salmonella enterica, Listeria monocytogenes and Campylobacter jejuni | 1 × 10-4ng for each genomic DNA | Naturally contaminated fresh meat samples: chicken, beef, pork and turkey | Not stated |
| Listeria monocytogenes | 8 log CFU/mL | Artificially contaminated milk | Not stated | |
| Escherichia coli, Shigella spp., Salmonell spp., Proteus spp., Campylobacter jejuni, Enterococcus faecalis, Yersinia enterocolitica, Vibrio parahaemolyticus, Vibrio fluvialis, ß-hemolytic streptococcus, Staphylococcus aureus | 10 CFU/mL of pure culture | Artificially and naturally contaminated pork, chicken, fish and milk | Not stated |
Table 4: Detection of Food Borne Pathogens using conventional methods, histological and radiological techniques.
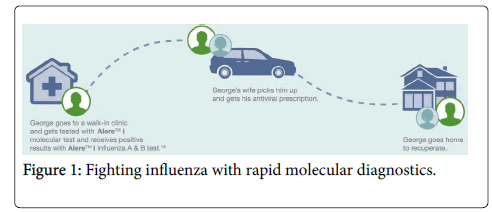
Let us look at the quick look at George’s case where both a timely molecular test enables him to go home within a matter of hours.
Accurate Diagnosis of Infectious Diseases
Without no doubt, the use of conventional methods to study infectious diseases was a success, however, a more substantial contribution came into reality when microbial pathogenicity at the molecular level comes in [15,18]. This has created a wider understanding of clinical manifestations, diagnosis, treatment and immunoprophylaxis of infectious diseases [15].
Since traditional methods depend on morphologic and metabolic features, they often lack the ability to show the genetic diversity found among the strains [15] and this is something that Nucleic acid Amplification Techniques (NAT) excels at. NAT are the leading methods in molecular diagnosis. They are causing a big impact on the study of microbial pathogenesis and even on the infectious diseases diagnosis [15-17]. Epidemiology always cannot precede the onset of an outbreak, and before an outbreak can be recognized, the circulation of diseases agent in a population can easily be detected with molecular methods [15]. In the below study, the table compared the accuracy of the diagnosis with multiplex PCR and culture using both direct and enriched methods.
Demistifying the Cost Involved in Patient Treatment
Being hospitalized costs money, whether paid for by the government, some insurance ageny, or the individual, receiving a treatment has never been an economic treat but being hospitalized and paying for the nights-stay has always been a problem. The conventional methods although reliable enough for diagnostic, require hospitalisation before the results can be obtained. For example if a patient whose sample will be analysed using conventional methods will stay for 3 nights that will be paid at $50 per night and another $50 for the test. This patient can pay $150 dollars for molecular detection and go home the very day. To minimize the hospitalized or turnaround period, automated machines are able to send results immediatly to the information system through an interface [3,12]. In the recent past after PCR was introduced in the microbial diagnostic laboratories, most technicians thought that the costs involved cannot be sustainable. However, this is similar to the law of demand and supply in markets, with the mass production of software applications, primers and probe designs, the cost of molecular methods falls [10]. For example with nucleic acid amplifications (NAAs), there is an increase in the use of these techniques in the diagnostic laboratories [8,10,12,17]. NAA with regards to the attainment of high-quality patient care seems to be the choice since it is fairly reasonable to be performed in the microbiology laboratory [10,13]. Respiratory pathogens have been proved quicker to diagnose when using molecular methods than traditional methods [10,11].
Non-viable Culture Organisms (meaning)
Molecular methods have come as a blessing to the clinical microbiology laboratories with the slogan ‘impossible is now more possible’. In the last decades, organisms that cannot be grown in culture or that are very difficult to grow for example; Hepatitis B virus (HBV), Hepatitis C virus (HCV), Human Immuno Deficiency Virus (HIV), Ebola Virus (EBV) and Cytomegalovirus (CMV) can now be efficiently detected with the use of PCR methods in a matter of hours [1]. Also due to the infectious dose, highly infectious agents that cannot be considered for culture. For example; Francisella tularensis, Brucella Spp, Coccidioides immitis etc. can now be identified through the molecular methods [1]. With the invention of molecular methods, a greater depth of knowlege is known about some critical organisms (fastidious) that require specialized media or culture conditions to grow [12,17]. In the recent discoveries, the information which said that only a few bacteria can be grown on artificial media has saved us from what we have been missing in specimen diagnostic. Now with culture-independent methods, both diagnostic and study of disease pathogenesis has been greatly acknowledged [12,18].
The Need to Wipe off the Tears of Developing Nations
The word ‘developing’ itself is challenging. Many of these nations that carries the big burden of the long historic malaria, tuberculosis and HIV and many other infectious agents cannot afford molecular methods in clinical diagnosis [2]. Usually these are due to the cost involved and the technical expertise that are unaffordable, as of today [2]. The monitoring of progression and the control of the spread of these dreadful resistant pathogens can only be accomplished with the molecular methods [2].
Our Big Hopes for the Future
The rapid detection and quantification of microbial pathogens in the clinical laboratories have exceedingly been recognised. With the introduction of PCR in clinical diagnostic laboratories, the detection and quantification of infectious microorganims, genetic disorder and cancer cells has gained the upper hand [1,3]. PCR is one-tube system that minimizes the risk of contamination. It can amplify long target up to 6.0 kb and also offer high sensitivity and can be reproducible analysis of both DNA and RNA [18]. PCR is the most popular approach in the family of diagnostic molecular methods and it has wiped out the need for extensive specimen preparation and improved automation [3]. Multiple pathogens can be detected simultaneously with the use of multiplex platforms [3].
In short, multiplex PCR has the ability to detect number of potential pathogens simultaneously. The coming of the high-throughput sequencing (HTS) to genetics, has made a significant revolution in the diagnosis of infectious diseases. Henceforth there is a need for these methods to replace the conventional methods in the diagnostic laboratories [5].
We are hopeful that with the use of NGS the discovery of novel pathogens that might be related to acute or chronic illnesses will be achieved. Appropriate quality controls, interpretation of data and the fullfilment of regulatory requirements are considered to be crucial [13]. Direct sequencing and also microarray analysis will lead the molecular diagnosis with great hopes in both research and clinical laboratories [10]. Below are the already molecular methods but limited to, that have shown the diagnostic laboratories.
The Limitations of Molecular Methods
In the health departments and especially in the clinical diagnostic sections, a serious patient care is always needed, because a single mistake made during the diagnosis can lead to a life being lost. In this regard, despite the nobility of our faithful molecular methods in the diagnostic laboratories, we still have to cross-check its limitations.
One of the limitations is based on the species whose gene has not been included in the genome database known. Molecular methods lack the detection of this species and cannot detect if there is a newly emerging resistance mechanisms [13,16].
Second to the emergence of new genes is the unbalance nature of the microbiology laboratories between the developed and developing nations, and the non-bridging of microbiology and molecular laboratories [2].
A matter of hours can enable results or us to gain and hence treatment is halfway completed. However, a typical DNA sequencing is done over night, whereas s quick blood cultivation test can be done in a matter of hours [12].
While the accurate and advanced molecular laboratory techniques contributed to a more effective diagnosis, patient care and management in developed nations, the developing nations are still struggling with the only available ill equipped and sometimes the undertrained personnel [2,12]. It requires a concommitant techniquical know-how [12,16].
Majority of the availlable test kits are typically limited to the detection of one or a few pathogens [3,16] and this means more different kits are needed for diagnosing various agents. This is exponentially expensive. Of course, the margins between the cost of conventional and molecular diagnostic test is enough for one to go in for conventional tools especially in the clinics where numbers of sample are waiting on the bench. In addition, molecular methods are a lot more expensive than traditional methods, as seen from the healthcare system’s point of view. Sending a sampe off for DNA sequencing will produce an invoice that has to be paid. However, using the traditional approach, there are no invoices (as the healthcare systemsees it), just staff doing the culturing/microscopy etc. [3]. Still on the cost involed, the fact that prolonged hospitalization also costs money, and that staff needs salary, is not factored in as a cost for tradition methods since there are no invoices for these. In short from where a healthcare system is looking, molecular methods for microbiological identification are extremely expensive (the traditional methods, after all, are for free). In some cases, molecular methods only augment cultures like; where live bacteria is needed to conduct antimicrobial susceptibility testing (AST) [3,16]. Aside from mecA (a gene found in bacterial cells that enable resistance of bacteria to antibiotics like penicillin and its likes) and VanA (a gene responsible for vancomycin-resistant), AST assays are very few at hand for specific genes [3].
Contamination of all kinds also joins in the factors of limiting the use of molecular methods in diagnostic laboratories. One of these is the carryover contamination [12]. The pre and post amplification contaminants always translated into false positivity [12,16].
Human imperfections that bring in sampling error also inhibit the assay, causing false negatives [12]. Traditional methods will still take a lead for the reasons of being tradition. Since we always have the believed that ‘this is how we do it’ and we will more or less prefer to be doing it like this. For example if salmonella could be culture and identified by biochemical test, going futher with confirmation with molecular tests might be less on the option [16]. However if all the laboratory technicians are well sensitived on the emergence of molecular techniques and the coniderble reduction of the cost involved, this hurdle can be overcome.
Molecular methods when compared to conventional approaches seems more expert oriented. One example is a test that involves the use of 16S rRNa gene amplification and sequencing [10]. This is the best for bacteria if the purity of sample is assured; however, it is always a hurdle if the corresponding culture is negative. Probably there are mixed infections or unusual pathogens [10]. In addition, deviant bacteria are likely to require very specialized PCR primersç; if you just use regular bacteria, you will miss them [5].
Conclusion
Clinical microbiology laboratories in the developing nations should be updated with the recent molecular techniques while revolutionized to add a more efficient diagnosis and patient care. Previous studies have revealed that severe morbidity and mortality are due to the lack of point care accurate laboratory diagnostic methods. In this response, the introduction of more sophisticated molecular methods will effectively help in the control of infectious disease epidemics and pandemics that are of public health importance. In fact, it is time for for the healthcare system to take the step over to molecular methods in, for example bacterial identification.
The accreditation of laboratories and competence of the staff in molecular diagnostic laboratories should be frequently monitored.
In addition to the need of standard protocols or standard operating procedures, reference materials and proficiency testing programmes for testing methods will give results that are more consistent among different laboratories.
With the implementation of Good Clinical Laboratory Practices (GCLP) or Health and Safety for example handwashing, reporting of compliance and with regular monitoring and surveillance of handwashing will drastically help to reduce nosocomial infections [6].
References
- Fournier PE, Drancourt M, Colson P, RolainJM, Scola BL, et al. (2013) Modern clinical microbiology: new challenges and solutions. Nat Rev Microbiol 11: 574-585.
- Muralidhar S (2015) Molecular methods in the laboratory diagnosis of sexually transmitted infections. Indian J Sex Transm Dis 36: 9-17.
- Wilson LM (2015) The Accelerating Transition From Culture-Based to Molecular-Based Methods. Am J Clin Pathol 143: 766-767.
- CoboF (2012) Application of Molecular Diagnostic Techniques for Viral Testing. Virology J 6: 104-114.
- Hodkinson BP, Grice EA (2015) Next-Generation Sequencing: A Review of Technologies and Tools for Wound Microbiome Research. Adv Wound Care (New Rochelle) 4: 50-58.
- Sohn AH, Garrett DO, Sinkowitz-Cochran RL, Grohskopf LA, Levine GL, et al. (2001) Prevalence of nosocomial infections in neonatal intensive care unit patients: results from the first national point-prevalence survey. J Pediatr 139: 821-827.
- Cheng JW, Xiao M, Kudinha T, Kong F, XuZP, et al. (2016) Molecular Epidemiology and Antimicrobial Susceptibility of Clostridium difficile isolates from a University Teaching Hospital in China. Front Microbiol 7: 1621.
- Malhotra S, Sharma S, Bhatia NJK, Kumar P, Bhatia NK, et al. (2014) Recent Diagnostic Techniques in Mycology. J Med MicrobDiagn 3: 146.
- Babouee B, Goldenberger D, Elzi L, Lardinois D, Sadowski-Cron C, et al. (2013) Prospective study of a panfungal PCR assay followed by sequencing, for the detection of fungal DNA in normally sterile specimens in a clinical setting: a complementary tool in the diagnosis of invasive fungal disease? Clin Microbiol Infect 19: E354-357.
- CLSI (2011) Establishing molecular testing in clinical laboratory environments; draft guideline. CLSI document MM19A. Clinical and Laboratory Standards Institute, Wayne, PA.
- Ginocchio CC, McAdamAJ (2011) Current Best Practices for Respiratory Virus Testing. J Clin Microbiol 49: S44-S48.
- Persing DH, Fred TC, Yi-Wei T, Frederick NS, Randall HT, et al. (2014) Molecular microbiology: diagnostic principles and practice 2nd ed. Lab Med 45: e8.
- Hryniewiecki T, Gzyl A, Augustynowicz E, Rawczynska-Englert I (2002) Development of broadrange polymerase chain reaction (PCR) bacterial identification in diagnosis of infective endocarditis. J Heart Valve Dis 11: 870-874.
- Satish G, Ravikanth, Manideep K (2016) Role of Molecular Epidemiology in Infectious Disease Surveillance. J Hum Virol Retrovirol 3: 00111.
- Oliver DM, van Niekerk M, Kay D, Heathwaite AL, Porter J, et al. (2014) Opportunities and limitations of molecular methods for quantifying microbial compliance parameters in EU bathing waters. Environ Int 64: 124-128.
- Clinical and Laboratory Standard Institute (CLSI) Molecular Diagnostics Methods for Infectious Dieseses; Aproved Guidline Second Edition. CLSI documents 31.
- Ramana KV (2014) Molecular Diagnostic Methods and Their Application to Patient Care: Clinical Microbiologist’s Perspective. Am J Clin Med Res 2: 8-13.
- Verweij JJ, Stensvold CR (2014) Molecular Testing for Clinical Diagnosis and Epidemiological Investigations of Intestinal Parasitic Infections.Clin Microbiol Rev 27: 371-418.
Copyright: © 2017 Bajinka O, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.